fro these processes, entropy change is positive.Ģ)Reaction taking place between a piece of marble or sodium hydroxide and Hydrochloric acid in an open vessel. These processes are accompanied by increase of randomness and hence increase of entropy i.e. These processes do not involve any exchange of matter and energy with the surrounding.Hence they are isolated systems. If we compress the gas isothermally from volume V 2 to V 1, heat q rev will be given out by the system and absorbed by the reservoir so that ΔS = − q rev / T and ΔS = q rev / T Total change in entropy ΔS 1 = ΔS sys + ΔS res = q rev / T + ( − q rev / T ) =0 The system absorbs heat q isothermally and reversibly at temperature T and expands from volume V 1 to V 2.Īs equivalent amount heat is lost by the reservoir, 
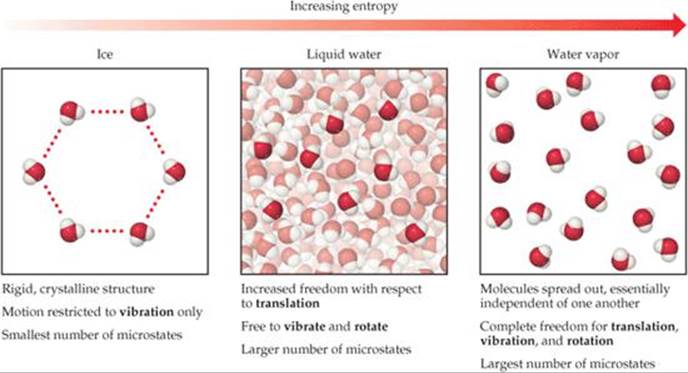
The physical significance of entropy is that many processes which are accompanied by an increase of entropy are also accompanied by an increase of randomness or disorder.Ĭonsider a system consisting of a cylinder containing a gas at fitted with frictionless and weightless piston and placed in contact with the large heat reservoir. The units of entropy change are cal/K/mol in CGS system and joules/K/mol in S.I. This shows that entropy change is inversely proportional to temperature.Įntropy change during a process is defined as the amount of heat ( q ) absorbed isothermally and reversibly divided by the absolute Temperature ( T ) at which the heat is absorbed. More the heat absorbed ,greater is the disorder.Ģ) For the same amount of heat absorbed at low temperature, the disorder is more than at high temperature. ΔS = S 2 -S 1 = ∑S products – ∑S reactantsġ) When a system absorbs heat ,the molecules start moving faster because kinetic energy increases. The change in its value during a process, is called the entropy change. Solid state has the lowest entropy, the gaseous state has the highest entropy and the liquid state has the entropy in between the two.Įntropy is a state function. The greater the randomness, higher is the entropy. Finally, an outlook, prospects and Quo Vadis of this field provides guidance for the development of new and improved HE-CCs and their derivatives to pave the way for future advancements.Entropy is a measure of randomness or disorder of the system. Besides, we describe effective and promising strategies for rationally designing HEMs derived from HE-CCs. Herein, the progress of the recently developed HE-CCs, including synthesis strategies and their performances for catalyzing the hydrogen evolution reaction (HER) and oxygen evolution reaction (OER) in water electrolysis as well as electrochemical performances in rechargeable batteries are highlighted. In fact, high-entropy coordination compounds (HE-CCs) have been considered a new family for the next generation of electrode materials in energy technologies. Recently, the library of HEMs was further expanded, and the concept of high entropy was introduced into some coordination compounds such as metal–organic frameworks (MOFs), metal-glycerate/metal-glycerolates as well as metal-polyphenol coordination polymers which may facilitate new opportunities for targeted applications. High-entropy materials (HEMs) have attracted great attention due to the distinctive designing concept and unique properties which are expected to make new advances in several energy conversion and storage technologies. 
0 Comments
Leave a Reply. |
AuthorWrite something about yourself. No need to be fancy, just an overview. ArchivesCategories |

 RSS Feed
RSS Feed
